calsfoundation@cals.org
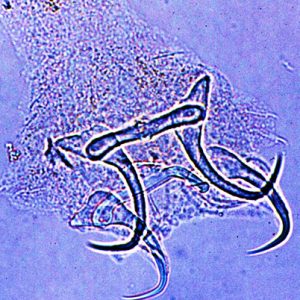
Monogeneans
The class Monogenoidea is a fairly large group of parasitic flatworms belonging to the phylum Platyhelminthes. Monogeneans are generally found on bony fishes in freshwater and marine habitats. Although some are endoparasites in the urinary bladder and eyes, most monogeneans are ectoparasites that attach to their host’s skin or gills by a special posteriorly positioned attachment organ called a haptor. The class contains about nine orders, fifty families, and 4,000–5,000 species. The genus Dactylogyrus is one of the largest genera, with nearly 1,000 species. Some taxonomists divide the Monogenoidea into two or three subclasses based on the complexity of their haptor. Those in the subclass Monopisthocotylea have one main part to the haptor, often with hooks or a large attachment disc, whereas those in the subclass Polyopisthocotylea have multiple parts to the haptor, typically in the form of clamps. Classification of the Monogenoidea is not universally accepted, and some of the taxonomy is controversial.
Several species occur in Arkansas, with several types of monogeneans native to the state. The monogeneans known as gyrodactylids are economically important as parasitic pests on bluegills, goldfish, and trout. Arkansas has thousands of farm ponds, but, by 2017, only one study had been done to determine the effect of monogenean parasites on Arkansas aquaculture. The study reported the seasonal incidence of several types of monogeneans in Arkansas fish farms but did not report on any specific mortalities or other effects. In general, it seems that only very heavy infestation causes mortalities.
Danish naturalist Otto Friedrich Müller (1730–1784) is thought to be the first (in 1776) to have described a monogenean; he collected it from the skin of halibut (Hippoglossus hippoglossus), and, thinking it was a leech (species Hirudo), named it Hirudo hippoglossi. It was not until 1858 that its status as a monogenean was established by Pierre-Joseph van Beneden (1809–1894); he named it Epibdella (now Entobdella) hippoglossi. He further published a detailed and accurate description of the parasite and produced some excellent illustrations. It is one of the largest monogeneans, measuring up to two centimeters in length. It has a smaller relative, measuring five to six millimeters in length, which was described by van Beneden and Hesse in 1864 and named Phyllonella (now Entobdella) soleae from the skin of the dover or common sole, Solea solea. This parasite is now perhaps the best understood monogenean.
The first overview of monogenoideans was by M. Maximilian Gustav Christian Carl Braun (1850–1930) from his investigations on the group between 1889 and 1893. The Swiss parasitologist Otto Fuhrmann (1871–1945) helped establish the Monogenea as a taxon separate from—but closely allied to—the Digenea in 1928. However, it was Boris Evseevitch Bychowsky (1908–1975), who, in 1937, was the first to propose Monogenoidea as a separate class apart from but equal to the Digenea. More recently, several papers on fish monogeneans have been published by Donald G. Cloutman and Delane Kritsky, both of whom continue research on this group of parasites, with Cloutman performing research in Arkansas.
Monogenean ancestors were probably free-living flatworms similar to modern rhabdocoel turbellarians. An early evolutionary hypothesis was that these gave rise to the monogeneans, and these, in turn, gave rise to digeneans, from which the cestodes (tapeworms) were derived. Another view was that the rhabdocoel ancestor gave rise to two lines; one was monogeneans, which gave rise to digeneans, and the other line was cestodes. Modern phylogenetic analyses of the Platyhelminths using a molecular approach have revealed that monogeneans are more closely related to tapeworms than to trematodes.
In general, monogeneans are mostly found on a wide variety of bony fishes in freshwater and marine habitats. Every major group of fish-like vertebrate harbors monogeneans. Monogeneans are also widespread on cartilaginous fishes such as sharks and rays. The polystomatid monogeneans have colonized more advanced vertebrates, including salamanders, frogs, and freshwater turtles. Surprisingly, one mammal, the hippopotamus (Hippopotamus amphibious), harbors Oculotrema hippopotami in its eyes. As far as is known, there are no monogeneans that infect snakes, lizards, or birds. Most monogeneans are browsers that move about freely on the fish’s body surface, feeding on mucus and epithelial cells of the skin and gills; however, a few adult monogeneans will remain permanently attached to a single site on the host. Some monogenean species invade the rectal cavity, ureter, urinary bladder, body cavity, and even the blood vascular system.
The polyopisthocotyleans almost exclusively dwell in gills and eat blood and tissue, whereas monopisthocotyleans can live on fishes’ gills, skin, or fins. A group of mostly ectoparasitic worms contains a few species that are endoparasites in the eyes, urinary bladder, or cloaca of vertebrate hosts. The parasitic monogeneans have complicated attachment organs at the posterior or tail end of their bodies, often including a mixture of suckers, clamps, hooks, and spines. The anterior structures are collectively termed the prohaptor, while the posterior ones are collectively termed the opisthaptor (or haptor) with its clamps, anchors, or hooks. This is usually the significant attachment organ.
Monogeneans do not have circulatory, respiratory, or skeletal systems and have a weak oral sucker—or none at all. As is the case with other flatworms, monogeneans have no true coelom (body cavity). Most monogeneans are covered with a tegument (a cluster of proteins) that is often supplied with short, scattered protrusions from the cellular membrane called microvilli; however, in some species, microvilli are absent, and shallow pits occur. Superficial muscles immediately below the tegument are arranged in circular, diagonal, and longitudinal layers. Monogeneans also have a simple digestive system consisting of a mouth opening with a muscular pharynx and an intestine without an anus. Monogeneans are also triploblastic—that is, they possess three embryonic germ layers: the endoderm, mesoderm, and ectoderm. In addition, they have a head region that contains concentrated sense organs and nervous tissue. The general nervous system is composed of cerebral nerve cell clusters called ganglia in anterior and posterior nerve trunks. Their sense organs are variable; most have pigmented eyes in free-swimming larvae, and in many species the adults have eyes as well.
In terms of reproduction, all monogeneans are hermaphroditic, with the functional reproductive organs of both sexes present in a single individual; cross-fertilization usually takes place. The testis is single or follicular (up to 200/individual in one species); sperm are evacuated into a specialized, often sclerotized copulatory organ. Each testis has a vas efferens that fuses into an ejaculatory duct, without a cirrus pouch or eversible cirrus (meaning a pouch that can be turned inside out).
Several families of monogeneans have a complex copulatory organ composed of a sclerotized (hardened) apparatus that joins with the ejaculatory duct. This apparatus consists of an accessory piece and a male copulatory organ. These male reproductive structures are highly variable among monogeneans and have been found to be important taxonomic characters used to help differentiate between similar forms.
Female organs include an ovary (germarium) and follicular vitelline glands. The oviduct leaves the ovary and travels toward the oocyte (which will eventually become the zygote), receiving vaginal, vitelline, and genitourinary ducts. A seminal vesicle is often present with abundant vitellaria, usually extending throughout the parenchyma and sometimes even into the opisthohaptor. The uterus usually contains one or only a few eggs. However, South American oviparous gyrodactylids may have up to twenty intrauterine ova; their uterus contains prenatal offspring, and vitelline follicles are absent.
Following fertilization, an oocyte and attendant vitelline cells pass into the ootype, a muscular expansion of the female duct. In some species, a Mehlis’ gland around the ootype comprises two cell types. The function of this gland is not known. Although several eggs can be produced by some species of monogeneans, Polystoma spp. can shed one to up to three eggs every ten to fifteen seconds. Some monogeneans can store a few eggs in the ootype and then eject them to the outside directly through a pore; however, in most species, the eggs pass from the ootype into a uterus.
Monogeneans possess a relatively simple, single life cycle compared to other parasitic worms. It is direct (they have no intermediate hosts), and although they are hermaphrodites, the male reproductive system begins to function before the female part does. When the eggs hatch, it releases a heavily ciliated (fringed) larval stage called an oncomiracidium. The oncomiracidium has many posterior hooks and is generally the life stage responsible for host-to-host transmission. These larvae swim until they contact a suitable host where they attach, lose their ciliated cells, and develop into an adult.
However, there is an exception to this reproductive plan—the viviparous monogeneans of the family Gyrodactylidae. They are unusual among the monogeneans as they exhibit sequential polyembryony, with as many as four individuals resulting from a single zygote. In not having an oncomiracidium, gyrodactylids must depend on transmission of the subadult or adult from one host to another. These unciliated forms appear unable to swim, so they must be in close proximity to the worm’s current host for transfer to take place successfully or establish themselves on the same host as their parents (called autoinfection). Indeed, infections seem to spread easily in fish hatcheries, as the worms can leave a dead host and remain active for several hours. Even when infected salmon with gyrodactylids were removed from a fish tank, uninfected eels replaced in the same tank became infected within the next day.
Although Arkansas has over 127,000 farm ponds, only one study, as of 2017, has been done on the effect of monogenean parasites on Arkansas aquaculture. The study reported the seasonal incidence of Dactylogyrus, Gyrodactylus, and Ligictaluridus (reported as Cleidodiscus) in Arkansas fish farms but did not report on any specific mortalities or other effects. It also noted that Ligictaluridus normally does not cause mortalities on catfish in fish farms. On the other hand, in another study, mortality was reported that was due to an extremely heavy infestation of Ligictaluridus pricei in a catfish culture pond in Alabama with high stocking density (2,000/acre).
Epidemics and excessive parasite loads of monogeneans on captive and wild fishes occur around the world. For example, Gyrodactylus salaris is a notorious killer in Norway on Atlantic salmon parr (Salmo salar), both in the wild and in fish farms. Fishes infested with monogeneans may become sick and lethargic, swim near the surface, have clamped fins, or inhabit the corners of fish tanks or the sides of fish farm ponds. Secondary infections are common on tissue damaged by these worms. As such, the best way to manage them is to avoid their introduction to a new aquatic system. There are several methods of elimination, including quarantine, chemical treatments, and drying/disinfecting tanks; however, it is very difficult to eliminate monogeneans completely once they are introduced.
In Arkansas, researchers are learning more about fish monogeneans. Several species occur in the state, including those in the genera Actinocleidus, Aethycteron, Acolpenteron, Anchoradiscus, Clavunculus, Cleidodiscus, Dactylogyrus, Gyrodactylus, Haplocleidus, Ligictaluidus, Octomacrum, Onchocleidus, Pellucidhaptor, Pterocleidus, Salguginus, and Syncleithrium. The following fish monogeneans are endemic in the state: Dactylogyrus asper in Benton County; D. boopsi in Franklin, Fulton, Newton, Polk, and Washington counties; D. greenei in Franklin, Johnson, and Perry counties; D. latrictus in Izard County; D. robisoni in Calhoun County; and Gyrodactylus bretinae in Clay County. More recently, monogeneans have been reported from mosquitofish and fundulid (topwater) fishes in Arkansas as follows: Salsuginus fundulus, S. seculus, and S. umbraenis. A new species of Aethycteron was reported from the sunburst darter (Etheostoma mihileze) from the Ozarks of Arkansas in Benton and Madison counties.
Little is known about amphibian monogeneans in Arkansas. One species, Sphyranura euryceae, was reported from the external gills, skin, and gular folds of larval and neotenic salamanders (Eurycea spp.) from Marion County. The same monogenean was found on the gills of larval grotto salamanders (Eurycea spelaea) and neotenic Oklahoma salamanders (Eurycea tynerensis) in adjacent northeastern Oklahoma. As of 2017, nothing has been published on monogeneans occurring in or on frogs and toads of Arkansas, although one genus (Polystoma) may eventually be found in these potential hosts. In nearby Oklahoma, two anuran polystomes, Pseudodiplorchis americanus, are known from the urinary bladder of spadefoot toads (Scaphiopus spp.), and Neodiplorchis scaphiopi infects the urinary bladder of the plains spadefoot toad, Spea bombifrons. Three species of spadefoots occur in Arkansas, and these monogeneans may eventually be found in their urinary bladders.
Concerning turtle monogeneans, nothing has been published as of 2017 on Arkansas hosts. In Texas, Polystomoidella whartoni was documented from the urinary bladder of a yellow mud turtle, and in Oklahoma, Neopolystoma sp. was found in the conjunctival sacs of a snapping turtle. With additional surveys on Arkansas’s seventeen species and subspecies of aquatic turtles, these two monogeneans, as well as others, may ultimately be found when examining the urinary bladder and eyes of these hosts.
For additional information:
Allison, R., and W. A. Rogers. “Monogenetic Trematodes of Some Alabama Freshwater Fishes with Descriptions of Four New Species and Redescriptions of Two Species.” Proceedings of the Helminthological Society of Washington 37 (1970): 17–23.
Bakke, Tor A., P. D. Harris, and J. Cable. “Host Specificity Dynamics: Observations on Gyrodactylid Monogeneans.” International Journal for Parasitology 32 (2002): 281–308.
Bakke, Tor A., P. D. Harris, P. A. Jansen, and L. P. Hansen. “Host Specificity and Dispersal Strategy in Gyrodactylid Monogeneans, with Particular Reference to Gyrodactylus salaris (Platyhelminthes, Monogenea).” Diseases of Aquatic Organisms 13 (1992): 63–74.
Boeger, Walter A., and Delane C. Kritsky. “Phylogeny and a Revised Classification of the Monogenoidea Bychowsky, 1937 (Platyhelminthes).” Systematic Parasitology 26 (1993): 1–32.
Buchmann, Kurt, and J. Bresciani. “Monogenea (Phylum Platyhelminthes).” In Fish Diseases and Disorders, Vol. 1: Protozoan and Metazoan Infections, 2nd ed. Edited by P. T. K. Woo. Oxfordshire, UK: Cab International, 2006.
Chien, Shih M. “Dactylogyrids from North American Cyprinids of the Genus Nocomis. The bellicus Group.” Journal of Parasitology 60 (1974): 585–594.
Cloutman, Donald G. “Dactylogyrus boopsi (Monogenea: Dactylogyridae) from the Bigeye Shiner, Notropis boops (Gilbert) (Pisces: Cyprinidae).” Journal of the Helminthological Society of Washington 61 (1994): 219–220.
———. “Dactylogyrus greenei (Monogenea: Dactylogyridae) from the Wedgespot Shiner, Notropis greenei Hubbs and Ortenburger (Pisces: Cyprinidae).” Journal of the Helminthological Society of Washington 62 (1995): 10–12.
———. “Dactylogyrus robisoni n. sp. (Monogenoidea: Dactylogyridae) from the Bluehead Shiner, Pteronotropis hubbsi (Bailey and Robison), 1978 (Pisces: Cyprinidae).” Comparative Parasitology 78 (2011): 1–3.
Cloutman, Donald G., and Chris T. McAllister. “Aethycteron robisoni n. sp. (Monogenoidea: Ancyrocephalidae) from the Sunburst Darter, Etheostoma mihileze Mayden (Perciformes: Percidae).” Systematic Parasitology 94 (2017): 145–150.
Du Preez, Louis H., Olivier Verneau, and Timothy S. Gross. “Polystoma floridana n. sp. (Monogenea: Polystomatidae) a Parasite in the Green Tree Frog, Hyla cinerea (Schneider), of North America.” Zootaxa 1663 (2007): 33–45.
Foster, W. D. A History of Parasitology. Edinburgh: E. & S. Livingston, 1965.
Hoffman, Glenn L. Parasites of North American Fishes, 2nd ed. Ithaca, NY: Comstock Publishing, 1999.
Kearn, Graham C. “Monogeneans, the Ultimate Fish Parasites.” Biologist 58 (2011): 28–32.
———. “Some Aspects of the Biology of Monogenean (Platyhelminth) Parasites of Marine and Freshwater Fishes.” Oceanography 2 (2014): 1–7.
Llewellyn, J. “Larvae and Larval Development of Monogeneans.” Advances in Parasitology 1 (2011): 287–327.
McAllister, Chris T., and Donald G. Cloutman. “Salsuginus seculus (Monogenoidea: Dactylogyrida: Ancyrocephalidae) from the Western Mosquitofish, Gambusia affinis (Cyprinodontiformes: Poeciliidae): New Records for Arkansas, Kansas and Oklahoma.” Proceedings of the Oklahoma Academy of Science 95 (2015): 42–45.
McAllister, Chris T., Charles R. Bursey, and Matthew B. Connior. “Helminth Parasites (Trematoda, Cestoidea, Nematoda, Acanthocephala) of Herpetofauna from Southeastern Oklahoma: New Host and Geographic Records.” Proceedings of the Oklahoma Academy of Science 95 (2015): 125–134.
McAllister, Chris T., Charles R. Bursey, William F. Font, Henry W. Robison, Matthew B. Connior, Donald G. Cloutman, and Thomas J. Fayton. “Helminth Parasites of the Blackspotted Topminnow, Fundulus olivaceus (Cyprinodontiformes: Fundulidae), from the Interior Highlands of Arkansas.” Journal of the Arkansas Academy of Science 69 (2015): 135–138. Online at http://libraries.uark.edu/aas/issues/2015v69/v69a24.pdf (accessed September 1, 2020).
McAllister, Chris T., Charles R. Bursey, William F. Font, Henry W. Robison, Stanley E. Trauth, Donald G. Cloutman, and Thomas J. Fayton. “Helminth Parasites of the Northern Studfish, Fundulus catenatus (Cypriniformes: Fundulidae) from the Ouachitas and Ozarks of Arkansas, U.S.A.” Comparative Parasitology 83 (2016): 78–87.
McAllister, Chris T., Charles R. Bursey, Thomas J. Fayton, Donald G. Cloutman, Henry W. Robison, Matthew B. Connior, and Stanley E. Trauth. “Helminth Parasites of the Blackstripe Topminnow, Fundulus notatus (Cyprinidontiformes: Fundulidae), from Arkansas and Oklahoma, U.S.A.” Comparative Parasitology 83 (2016): 227–236.
McAllister, Chris T., Charles R. Bursey, Michael A. Steffen, Samuel E. Martin, A. L. Trujano-Alvarez, and Ronald M. Bonett. “Sphyranura euryceae (Monogenoidea: Polystomatoinea: Sphyranuridae) from the Grotto Salamander, Eurycea spelaea and Oklahoma Salamander, Eurycea tynerensis (Caudata: Plethodontidae), in Northeastern Oklahoma, U.S.A.” Comparative Parasitology 78 (2011): 188–192.
McAllister, Chris T., Charles R. Bursey, and Stanley E. Trauth. “New Host and Geographic Distribution Records for Some Endoparasites (Myxosporea, Trematoda, Cestoidea, Nematoda) of Amphibians and Reptiles from Arkansas and Texas, U.S.A.” Comparative Parasitology 75 (2008): 241–254.
McAllister, Chris T., Stanley E. Trauth, and Lawrence W. Hinck. “Sphyranura euryceae (Monogenea) on Eurycea spp. (Amphibia: Caudata), from Northcentral Arkansas.” Journal of the Helminthological Society of Washington 58 (1991): 137–140.
Mehlhorn, Heinz, ed. Encyclopedia of Parasitology. 4th ed. New York: Springer International Publishing, 2016.
Meyer, Fred P. “Seasonal Fluctuations in the Incidence of Disease on Fish Farms.” American Fisheries Society Special Publication 5 (1970): 21–29.
Wellborn, Thomas L., Jr. “Four New Species of Gyrodactylus (Trematoda: Monogenea) from Southeastern U.S.” Proceedings of the Helminthological Society of Washington 34 (1967): 55–59.
Whittington, I. D, and L. A. Chisholm. “Diseases Caused by Monogenea.” In Fish Diseases, Vol. 2, edited by J. C. Eiras, H. Segner, T. Wahli, and B. G. Kapoor. Enfield, NH: Science Publishers, 2008.
Chris T. McAllister
Eastern Oklahoma State College





Comments
No comments on this entry yet.