calsfoundation@cals.org
American Eel
The American eel (Anguilla rostrata) belongs to the order Anguilliformes and family Anguillidae. Common names include Atlantic eel, common eel, freshwater eel, silver eel, yellow-bellied eel, green eel, black eel, bronze eel, elver, whip, and easgann. This family includes about eighteen facultative catadromous species of eels. The American eel ranges from Greenland and Iceland and all the drainages of eastern North America along the Atlantic and Gulf slopes west to New Mexico and south to Venezuela and islands of the Caribbean and West Indies across a latitudinal range of 5 to 62° N. In North America, A. rostrata occurs inland from eastern Canada to the Great Lakes, in the headwaters of many Atlantic and Gulf slope rivers, and in the Mississippi River Valley as far inland as Kansas, Nebraska, South Dakota, Minnesota, Wisconsin, and Pennsylvania south to Mexico. The closest relative of A. rostrata is the European eel (Angullia anguilla), which inhabits a similar latitudinal range in western Europe, and the two species are found in sympatry in Iceland. Although A. rostrata and A. anguilla are two distinct species, they are capable of hybridizing with each other. Fossils of freshwater eels date at least to the Eocene (56 to 34 million years ago).
In Arkansas, this eel’s distribution is sporadic, but generally it is found in the larger rivers of the state. The American eel appears to be especially abundant in the lower Arkansas River, where anglers sometimes by chance catch them using live bait. Unfortunately, populations of A. rostrata have been harmed in the state by the construction of hydroelectric and other locks and dams, blocking migration on virtually all of the major rivers.
In general, eels possess elongate serpentine mucus-covered bodies and well-developed jaws; they lack pelvic fins, and some lack pectoral fins, while scales are either absent or rudimentary and embedded below the epidermis as cycloid-type. Its lateral line is well developed, its head is long and conical, and it has small eyes. The mouth is terminal with small pectinate or setiform teeth, and the gill openings are well developed. The American eel shares all of these characteristics as well as a rounded caudal fin confluent with the dorsal and anal fins. In coloration, there are several variations, from olive green, brown shading to greenish-yellow, and light gray or white below. Eels from non-turbid waters are often lighter than those from dark, tannic acid–stained streams. The pectoral girdle possesses seven to nine (up to eleven in the young) radial elements. The gill opening is on the side in front of the lower half of a well-developed pectoral fin; the lower jaw is longer than the upper, and there are 103 to 111 vertebrae.
In Arkansas, most records of A. rostrata have been based on specimens caught by local fishermen representing size classes ranging from 381 to 762 mm (15 to 30 in.) in length. American eels can grow to 1.2 m (4.0 ft.) in length and up to 7.5 kg (17.0 lb.) in weight; females are generally larger than males. The maximum reported length is at least 1.2 m (47.0 in.), and the all-tackle world-record American eel was caught in 1995 in New Jersey and weighed 4.2 kilograms (9.0 lbs., 4.0 oz.). The Arkansas rod and reel record was an A. rostrata caught in the Spring River in 2015 and weighed 2.5 kg (5.0 lbs., 4.4 oz.).
The American eel has an exceptionally complex life history and includes coastal, estuarine, freshwater, and oceanic environments. Anguillids are obligate warm-sea spawners that must reproduce in tropically influenced waters with currents suitably directed to return their larvae to mouths of coastal rivers. During autumn, sexually mature A. rostrata migrate to and spawn (once) far offshore across several hundred thousand square kilometers in the Sargasso Sea bounded by four currents near Bermuda at depths of 400 to 500 meters (0.2 to 0.3 miles) in the southern North Atlantic Ocean and then die. The eggs hatch within a week of deposition, with emerging young peaking in February and continuing until April. Each female can lay 500,000 to 4 million buoyant eggs a year; larger individuals can release as many as 8.5 million ova. Life history stages include the leptocephali, glass eel, elver, yellow eel, silver eel, and adult.
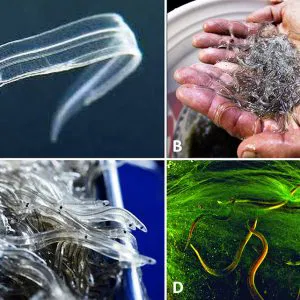
Leptocephali
After the eggs hatch, the early-stage young develop into leptocephali (a transparent marine stage) larvae with small, pointed heads and large teeth. These young eels passively move toward the eastern coast of North America for seven to twelve months. Studies suggest that leptocephali do not feed on zooplankton but rather ingest detrital particles such as marine snow (a continuous shower of mostly organic detritus falling from the upper layers of the water column) and fecal pellets or particles such as discarded houses of solitary free-swimming larvacean tunicates.
Glass Eels
As leptocephali enter the continental Gulf shelf, these 55 to 65 mm (2.1 to 2.6 in.) larvae metamorphose into glass eels (post-larval juveniles), which are transparent and possess the typical elongate and serpentine eel body form. Average age at this metamorphosis has been estimated at 200 days and estuarine arrival at 255 days. As they enter coastal waters, the animals essentially transform from a pelagic oceanic organism to a benthic continental organism.
Elvers
Glass eels become progressively pigmented as they approach the shore, becoming what are known as elvers, which feed primarily on insect larvae. During this three- to twelve-month phase of the life cycle, the young eel remains sexually immature. Once elvers enter freshwater, they may spend much of this period migrating upstream.
Yellow Eels
Once glass eels arrive on the continental shelf and enter freshwater systems, they grow as yellow eels (a sexually immature adult phase). In this phase, the eels are still primarily nocturnal. Sexual differentiation occurs during this stage and appears to be strongly influenced by environmental conditions. Yellow eels are essentially nocturnal benthic omnivores, and their prey includes bivalves, crustaceans, insect larvae, surface-dwelling insects, mollusks, worms, fishes, frogs, and plants.
Silver Eels
As the young yellow eels matures, it metamorphoses into a silver eel that takes on several anatomical and physiological attributes that allow it to migrate to the Sargasso Sea. Silver eels begin to migrate upstream and mature in rivers of eastern North America.
Eels are extremely mobile fishes and may access habitats that may otherwise appear unavailable to them by utilizing small watercourses or moving through wet vegetation. Small eels (<100 mm [3.9 in.]) in total length) are able to climb and may succeed in passing over vertical barriers. Adult eels have been reported to move overland and around obstacles such as dams on rainy nights. However, available habitat for eels has been reduced by habitat deterioration, barriers to upstream migration (for larger eels), and other barriers (e.g., turbines on hydroelectric dams) to downstream migration that can result in mortality. Indeed, dams on major rivers have severely limited the extent of the upstream migration, and, thus, the distribution of American eels. Interestingly, laboratory studies have demonstrated that eels are able to detect magnetic fields at different life stages.
When not out to sea or in brackish water, the American eel is mostly a bottom-dweller that inhabits rivers, muddy or silt-bottomed lakes, and permanent streams with continuous flow. During colder months, eels may burrow under the mud and enter a state of torpor (or complete inactivity) at temperatures below 5°C (41°F), although they might be occasionally active during this period. Similar to other anguillid eels, American eels are nocturnal hunters that use a keen sense of smell, and during the day they hide in burrows, tubes, snags, masses of aquatic vegetation, undercut banks, deep pools near logs and boulders, or at depths of about 1.5 to 1.8 meters (5.0 to 6.0 ft.) in mud, sand, or gravel very close to shore. They are generalist feeders on various aquatic organisms including larval insects (Ephemeroptera, Odonata, Plecoptera, Coleoptera, Trichoptera, and Lepidoptera), as well as gastropods, oligochaetes, amphipods, isopods, mysids, and fish from the families Anguillidae, Catostomidae, Cyprinidae, Ictaluridae, and Percidae.
Very little is known about the predators of eels, but elvers and small yellow eels are prey of large basses, although they are not considered a major part of their diet. All larval life stages are likely to be eaten by various predatory fishes. Older eels are also known to eat incoming glass eels, and, in turn, they also fall prey to other species of eels, bald eagles, and gulls, as well as other piscivorous birds.
Eels were once plentiful in rivers and were an important fish for indigenous people. Archaeological evidence indicates the existence of eel fishing for over 4,000 years. Adult A. rostrata are typically caught with eel pots and trot lines, whereas elvers and glass eels are caught with fine mesh fyke nets and dipnets. In various regions along the East Coast, they are considered economically important as bait for sport fishery such as that for the striped bass (Morone saxatilis), or rarely as a food fish in the United States. Although not considered a food fish in Arkansas, eels are an important commodity in some other regions of the world, particularly Europe and Japan, where smoked varieties and those baked in soy sauce are considered a delicacy. About ninety percent of freshwater eels consumed in the United States are now farm-raised but not bred in captivity. Instead, young wild eels are collected and then raised in various enclosures. However, several species used as “unagi doburi” (grilled eel rice bowl) have seen their population sizes greatly reduced in the past half century. Many populations in the wild are now threatened, and Seafood Watch of the Monterey Bay Aquarium recommend consumers avoid eating anguillid eels. In 2010, Greenpeace International included the American eel on its seafood red list. This list includes fish that are commonly sold in supermarkets around the world and that have a very high risk of being obtained from unsustainable fisheries. Most of the aquaculture eels are raised in Korea, Japan, and Taiwan, and with the greatest production in China.
In Arkansas, American eels are often observed in the Ouachita and White rivers, but perhaps due in part to fewer large river samples in the first two decades of the twenty-first century, recent observations are limited. In the early 1990s, sampling in the Ouachita River by the Arkansas Department of Environmental Quality produced twenty-nine specimens of A. rostrata. Just prior to 2000, penetration of eels into western Arkansas via upstream movement in the Arkansas River suggested that eels must have traversed several locks and dams; unfortunately, A. rostrata has not been collected there since 2001. In the 1980s, thousands of eels were reportedly observed in the Arkansas River below Wilbur D. Mills Dam No. 2 near Arkansas Post (Arkansas County). Additional fish surveys using various techniques (seines, trawls, and electrofishing) during the Arkansas River Navigation Study by personnel at the Environmental Laboratory, Engineer Research and Development Center (ERDC) in Vicksburg, Mississippi, produced 17,328 fishes representing sixty-five species, but not one A. rostrata was collected. Nonetheless, boat electrofishing in the lower Arkansas River in the vicinity of the post canal suggests the presence of a good eel population. Another study published in 2003 did not find any American eels in the lower Red River in Arkansas, although they were historically found there. However, that study did not include sampling using small-mesh hoop nets, and trotlines were used only on a limited basis. A commercial fisherman saw a single American eel in a fiddler net in the spring of 2005 in the Red River near Texarkana (Miller County). That method of capture may be limiting, however, as those nets were probably larger bar mesh, which capture only relatively large eels. In 2010, the drawdown of water to allow maintenance work on the dam in the lower lake and Caddo River (DeGray Lake watershed, Clark County) resulted in thirty-five A. rostrata (as well as other fishes) becoming trapped in a concrete rectangle below the spillway. Variation in size ranges observed in this sample of American eels likely represented multiple migrations from the sea, all of whose upstream progress was arrested by the dam.
Over the last half century, substantial decline in numbers and fishery landings of American eels over their range in North America (particularly Lake Ontario and the upper St. Lawrence River, Canada) has been noticed, raising conservation concerns over the status of this species. A sixty-five percent decline has been reported since the early 2000s in maturing eels from Lake Ontario and the St. Lawrence River area, and substantial declines have been observed in some areas of the Maritimes. Since the early 2000s, rapid declines of A. rostrata were recorded in Virginia, as well as in New Brunswick and Prince Edward Island in Canada. One major factor in eels being successful is their reproductive success as a catadromous species, which depends heavily on free downstream passage for spawning migration and on the availability of diverse habitats for growth and maturation. In addition, American eels are very sensitive to low dissolved oxygen levels, which is typically found below dams. Contaminations by heavy metals and other chemicals and pollutants can accumulate within the fat cells of eels, causing increased toxicity and reduced fecundity. But more so, the construction of dams and other irrigation facilities seriously decreases habitat availability and diversity for the eels in addition to hydroturbine mortality. Migration, population distribution, and prey availability can be affected by dredging as well as overfishing or excessive harvesting of juveniles that can also harm local populations. Management of the species has been virtually non-existent over the years. Information on the species is still limited, and much more effort is needed for a long-term plan to monitor localized populations. The International Union for Conservation of Nature (IUCN) Red List ranks the American eel as endangered. Its rounded overall global status is G4 (apparently secure) and in Arkansas is S3 (vulnerable) according to NatureServe; populations are critically imperiled (S1) in South Dakota and Ontario, Canada. Additional research is sorely needed on their current trend and the relative importance of the various threats on A. rostrata as well as other eel species of the world.
Many parasites have been reported from the American eel, including protozoans, monogeneans, trematodes, cestodes, nematodes, acanthocephalans, and crustaceans. An exotic, swim-bladder parasitic nematode (Anguillicoloides crassus) is a concern and is a native parasite of Japanese eels (Anguilla japonica) that spread from aquacultural imports from Asia. In North America, this nematode was originally discovered in a single American eel captured in South Carolina. Since then, the parasite has been detected in A. rostrata from different locations in the United States, including the Hudson River, Chesapeake Bay, Florida, Connecticut, and New Jersey. To date, however, nothing has been published on parasites of A. rostrata in Arkansas.
For additional information:
Barbin, G. P., and W. H. Krueger. “Behaviour and Swimming Performance of Elvers of the American Eel, Anguilla rostrata, in an Experimental Flume.” Journal of Fish Biology 45 (1994): 111‒121.
Bozeman, Earl L., Gene S. Helfman, and T. Richardson. “Population Size and Home Range of American Eels in a Georgia Tidal Creek.” Transactions of the American Fisheries Society 114 (1985): 821‒825.
Castonguay, Martin, P. V. Hodson, Catherine Couillard, M. J. Eckersley, J. D. Dutil and G. Verreault. “Why Is Recruitment of the American Eel, Anguilla rostrata, Declining in the St. Lawrence River and Gulf?” Canadian Journal of Fisheries and Aquatic Sciences 51(1994): 479‒488.
Cox, Casey Allen. “Population Demographics and Upstream Migration of American Eels in the Ouachita, White, and Arkansas Rivers.” MS thesis, University of Central Arkansas, 2014.
Denoncourt, Charles E., and Jay R. Stauffer Jr. “Feeding Selectivity of the American Eel Anguilla rostrata (LeSueur) in the Upper Delaware River.” American Midland Naturalist 129 (1993): 301‒308.
Dixon, D. A., editor. Biology, Management, and Protection of Catadromous Eels. American Fisheries Society Symposium 33. Bethesda, MD: 2003.
Dutil, J.-D., A. Giroux, A. Kemp, G. Lavoie, and J.-P. Dallaire. “Tidal Influence on Movements and on Daily Cycle of Activity of American Eels.” Transactions of the American Fisheries Society 117 (1988): 488‒494.
Helfman, Gene S., D. E. Facey, L. S. Hales Jr., and E. L. Bozeman Jr. “Reproductive Ecology of the American Eel.” American Fisheries Society Symposium 1 (1987): 42‒56.
Hill, Loren J. “Reactions of the American Eel to Dissolved Oxygen Tensions.” Texas Journal of Science 20 (1969): 305–313.
Hoffman, Glenn L. Parasites of North American Freshwater Fishes. 2nd ed. Berkeley: University of California Press, 2009.
Hodson, P. V., Martin Castonguay, Catherine M. Couillard, C. Desjardins, E. Pelletier, and R. McLeod. “Spatial and Temporal Variations in Chemical Contamination of American Eels, Anguilla rostrata, Captured in the Estuary of the St. Lawrence River.” Canadian Journal of Fisheries and Aquatic Sciences 51 (1994): 464–478.
Jonsson, B., and B. M. Jessop. “Geographic Effects on American Eel (Anguilla rostrata) Life History Characteristics and Strategies.” Canadian Journal of Fisheries and Aquatic Sciences 67 (2010): 326–346.
Kuwahara A., H. Niimi, and H. Itagaki. “Studies on a Nematode Parasitic in the Air Bladder of the Eel I. Descriptions of Anguillicola crassa sp. n. (Philometridea, Anguillicolidae).” Japanese Journal for Parasitology 23 (1974): 275–279.
Lee, David S. “Anguilla rostrata (LeSueur), American Eel.” In D. S. Lee, et al., Atlas of North American Freshwater Fishes. Raleigh: North Carolina State Museum of Natural History, 1980.
MacGregor, R., J. M. Casselman, W. A. Allen, T. Haxton, J. M. Dettmers, A. Mathers, S. LaPan, T. C. Pratt, P. Thompson, M. Stanfield, L. Marcogliese, and J. D. Dutil. “Natural Heritage, Anthropogenic Impacts, and Biopolitical Issues Related to the Status and Sustainable Management of American Eel: A Retrospective Analysis and Management Perspective at the Population Level.” American Fisheries Society Symposium 69 (2009): 713‒740.
Oliveira, Kenneth. “Life History Characteristics and Strategies of the American Eel, Anguilla rostrata.” Canadian Journal of Fisheries and Aquatic Sciences 56 (1999): 795‒802.
Ogden, J. C. “Relative Abundance, Food Habits, and Age of the American Eel, Anguilla rostrate (Lesueur), in Central New Jersey streams.” Transactions of the American Fisheries Society 99 (1970): 54‒59.
Richkus, William A., and Kevin Whalen. “Evidence for a Decline in the Abundance of the American Eel, Anguilla rostrata (LeSueur), in North America Since the Early 1980s. Dana 12 (2000): 83‒97.
Robison, Henry W., and Thomas M. Buchanan. Fishes of Arkansas. Fayetteville: University of Arkansas Press, 1988.
Rypina, Irina I., Joel K. Llopiz, Lawrence J. Pratt, and M. Susan Lozier. “Dispersal Pathways of American Eel Larvae from the Sargasso Sea.” Limnology and Oceanography 59 (2014): 1704–1714.
Sinha, V. R. P., and J. W. Jones. “On the Food of the Freshwater Eels and Their Feeding Relationship with the Salmonids.” Journal of Zoology 153 (2009): 119–137.
Tesch F.W. The Eel. 3rd ed. New York: Blackwell Science, 2003.
Tumlison, Renn and Henry W. Robison. “New Records and Notes on the Natural History of Selected Vertebrates from Southern Arkansas.” Journal of the Arkansas Academy of Science 64 (2010): 145‒150. Online at: https://scholarworks.uark.edu/cgi/viewcontent.cgi?article=1393&context=jaas (accessed July 11, 2019).
Walsh, P. J., G. D. Foster, and T. W. Moon. “The Effects of Temperature on Metabolism of the American Eel Anguilla rostrata (LeSueur): Compensation in the Summer and Torpor in the Winter.” Physiological Zoology 56 (1983): 532–540.
Wirth, Thierry, and Louis Bernatchez. “Decline of North Atlantic Eels: A Fatal Synergy?” Proceedings of the Royal Society B: Biological Sciences 270 (2003): 681–688.
Chris T. McAllister
Eastern Oklahoma State College






Comments
No comments on this entry yet.