calsfoundation@cals.org
Haemogregarines
Haemogregarines are protist parasites that belong to the Phylum Apicomplexa, Class Conoidasida, Order Eucoccidiorina, and Suborder Adeleorina, and include the following seven families: Adeleidae, Dactylosomatidae, Haemogregarinidae, Hepatozoidae, Karyolysidae, Klossiellidae, and Legerellidae. There are two major genera, Haemogregarina and Hepatozoon, in addition to several minor ones, including Babesiosoma, Bartazoon, Cyrilia, Dactylosoma, Desseria, Hemolivia, and Karyolysus. As a group, these parasites are cosmopolitan in their range of infected hosts (such as turtles, crocodiles, birds, reptiles, and mammals) and geographic distribution.
In general, haemogregarines sensu lato (meaning, in a broad sense) are elongate to fusiform ovoidal organisms that occur within the erythrocytes (red blood cells) of various animals, mostly poikilothermic vertebrates (animals whose internal temperatures vary widely). They form a group of diverse heteroxenous adeleid coccidian parasites that have exploited both terrestrial and aquatic environments, and, as such, infect numerous vertebrate hosts (turtles, crocodilians, birds, reptiles, and mammals). Four haemogregarine clades have been associated with acarine vectors (Hepatozoon, Karyolysus, Hemolivia) and another three with leeches (Dactylosoma, Desseria, Haemogregarina).
Many haemogregarines were originally described on the basis of gametocyte morphology and were most often designated as a Haemogregarina sp. or Hepatozoon sp. It is now known that observation of stages in the vector truly indicate the generic position of the parasite. Indeed, over the last century or more, nomenclature has been modified, while detailed information on life cycles has been documented. For example, Hemogregarina mauritanica, originally described in 1904, was renamed Hepatozoon mauritanicum in 1973 and Hemolivia mauritanica in 1997.
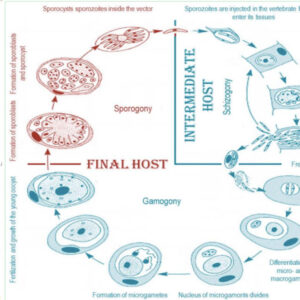
The life cycle of haemogregarines in aquatic hosts is transmitted by leeches or by arthropods in which the sexual part of the cycle develops. For example, there is a round of merogony after sporogony, and it is the merozoites that are infective from the leech bite. In terrestrial hosts, the life cycle of haemogregarines comprises these four stages: merogony and gamogony in the vertebrate host, and fertilization and sporogony in the invertebrate host. In addition to the normal life cycle in which transmission is achieved by a bite of a suitable vector or its ingestion by the vertebrate host, a second mode of transmission is utilized by some species, such as transmission by predation between vertebrates. This mode of transmission is shared by all species when the food habit of the host species does not include the direct ingestion of the vector by the vertebrate host.
The genus Haemogregarina was described in 1885 by Vasily Iakovlevich Danilewsky (1852–1939) from the European pond turtle (Emys orbicularis). He was the first to investigate systematically the blood parasites of amphibians, reptiles, and birds. Some other notable biologists who provided a great deal of early information on haemogregarines include Charles Louis Alphonse Laveran (1845–1922), Sir Edwin Ray Lankester (1847–1929), Johann Eduard Reichenow (1882–1960), Percy C. C. Garham (1901–1994), and Ralph Lainson (1927–2015). Some current notable biologists who have studied haemogregarines include Sherwin S. Desser of the University of Toronto; Mark E. Siddall of the American Museum of Natural History; Todd G. Smith of Acadia University, Canada; Sam R. Telford Jr., formerly of the Florida Museum of Natural History; and John R. Barta of the University of Guelph, Canada.
The family Haemogregarinidae includes members that are heteroxenous with gamonts within erythrocytes and/or other blood cells, and most have merogony in erythrocytes of a suitable vertebrate host. Sporozoites excyst within an invertebrate host and undergo merogony, and then merozoites are transmitted by feeding on the host by a leech vector. Although their size varies and different morphological forms exist, they are generally larger than the red blood cell nucleus. The majority of species have been described based on the morphology of their erythrocytic stagesand supposed strict host specificity.
Several species have been placed in the genus Haemogregarina. However, recent evidence suggests that only those species utilizing a leech definitive host may truly belong in this genus. Those in the genus Haemogregarina possess oocysts with eight sporozoites. These parasites infect lower vertebrates (fish and mainly reptiles) as intermediate hosts and leeches (as definitive hosts). Among reptiles, species of the genus Haemogregarina are known from turtles, lizards, and crocodilians.
One important Nearctic species is H. balli that possesses merozoites that migrate and accumulate in the salivary glands of leeches, Placobdella spp. The parasites enter the wound caused when a leech takes a blood meal from common snapping turtles, Chelydra serpentina. Merogony occurs in the turtle with immature meronts found in red blood cells, mature meronts with six to eight merozoites sequestered in the liver, and mature meronts with thirteen to twenty-five merozoites sequestered in the liver, lung, and/or spleen. The last generation merozoites form gamonts, which are found within erythrocytes.
The family Hepatozoidae includes heteroxenous members having gamonts within erythrocytes (and/or leukocytes) of amphibians, reptiles, birds, or mammals. Merogony occurs in vertebrate hosts, and transmission to these hosts is by ingestion of an invertebrate (e.g., mosquitos and other dipterans, bugs, mites, and ticks—and perhaps leeches in a few cases). These parasites have large polysporic oocysts with a distinct wall and form within tissues of an invertebrate definitive host. Sporocysts possess four or more sporozoites each. Some vertebrate host species harbor dormant monozoic or dizoic cystozoites in tissue cysts, especially in their lungs. In these cases, the cysts may be transferred to a third (vertebrate) host that is involved in the life cycle (i.e., paratenic-like host). The genus Hepatozoon, defined by morphology and the life cycle of its type species, He. perniciosum, has over the last century become a heterogeneous group of species with diverse life cycles; according to recent investigation, it is paraphyletic. Examples include He. catesbianae in American bullfrogs (Rana catesbeiana) that ingest mosquitos (Culex territans), He. canis in canids (Canis spp.), He. crotali in rattlesnakes (Crotalus spp.), He. griseisciuri in grey squirrels (Sciurus carolinensis), He. muris in rats (Rattus spp.) and mites (Laelaps echidninus), He. rarefaciens in Indigo snakes (Drymarchon corais) and mosquitos (several different genera), and He. thamnophis in garter snakes (Thamnophis spp.). In addition, there is He. clamatae, a parasite related to Plasmodium species that lives in the erythrocytes of green frogs (Rana clamitans) in North America. Some vertebrates require an intermediate (vertebrate) prey host harboring dormant monozoic or dizoic cysts in order to become infected. It is thought that these intermediate hosts undergo the same parasite development (i.e., merogony and gamogony) as the predator vertebrate host. For example, He. sipedon in water snakes (Nerodia spp.) cannot directly infect snakes when snakes are fed mosquitos harboring the sporulated oocysts; however, it utilizes frogs possessing dizoic hepatic cysts as the mode of infection. In addition, He. domerguei in snakes requires a specific lizard, Cuvier’s Madagascar Swift (Oplurus cuvieri), with dizoic lung cysts to become infected. However, the life cycle can occur between mosquitos and lizards without the need for snake hosts.
Similar to Hepatozoon, the genus Karyolysus was created in 1894 for those species of hematozoans that fragment the host nucleus. The type species is Karyolysus lacertae described from the European wall lizard (Podarcis muralis). Other hosts include lizards of the genera Darevskia, Lacerta, Mabouia, and Varanus. The vectors are gamasid mites from the genus Ophionyssus that are ingested by lizards. A molecular analysis of the 18s rRNA suggests that this genus may overlap with Hepatozoon.
The genus Babesiosoma, created in 1956, has two hosts in its life cycle. Vertebrate hosts are mainly fishes and some ranid frogs, and invertebrate vectors are leeches. The parasite produces four intraerythrocytic merozoites from cruciform meronts. However, compared to other hematozoans, this genus is poorly studied, and little is known about it.
Similar in life cycles with Babesiosoma, the genus Dactylosoma, described in 1894, includes species that have two hosts in their life cycle, a vertebrate host (fish or amphibian, and possibly reptile) and an invertebrate host (glossiphoniid leech). The type species is Dactylosoma ranarum, which occurs in ranid frogs. Dactylosomids are thought to occur in amphibians from Africa, North and South America, Asia, and Europe.
The genus Cyrilia was created in 1981 for hematozoans that infect freshwater fishes and is transmitted by the bite of African fish leeches, Batracobdelloides tricarinata. Hosts include the African catfish (Clarias lazera) and Nile tilapia (Tilapia nilotica).
The genus Desseria was described in 1995. Members were formerly considered to belong to the genus Haemogregarina. All forty currently recognized species in this genus infect fishes.
The genus Hemolivia was described in 1990 and the type species is H. stellata. This genus comprises tick-transmitted haemogregarines of ectothermic vertebrates. The vertebrate is the cane toad (Rhinella marina), and the invertebrate host is the tick Amblyomma rotondatum. Toads become infected by ingesting infected ticks. Other vertebrate hosts include blue-tongued skinks (Tiliqua rugosa), giant ameiva (Ameiva ameiva), Mediterranean spur-thighed tortoises (Testudo graeca), marginated tortoises (Testudo marginata), and ornate wood turtle (Rhinoclemmys pulcherrima manni). Several haemogregarines of amphibians and reptiles could probably be assigned to the genus Hemolivia if their life cycle were known.
The most recently created hematozoan genus is Bartazoon, described in 2015. The type species is B. breinli described from a freckled monitor lizard (Varanus tristis orientalis) from Queensland, Australia. This parasite is transmitted by biting insects.
The study of hematozoans in the state is in its infancy. Only Haemogregarina and Hematozoon have been reported from Arkansas turtles and snakes, respectively. To date, there are no reports of hematozoans from fishes, amphibians, birds, or mammals of the state. The first report of a hematozoan from any Arkansas host was in 1990 when a Haemogregarina sp. was found in the erythrocytes of red-eared sliders, Trachemys scripta elegans, collected from I. F. Anderson Minnow Farms in Lonoke County. Since then, there have been other reports of hematozoans as follows: Hepatozoon spp. from diamondback watersnake (Nerodia rhombifer), western rat snake (Pantherophis obsoletus obsoletus), and western ribbon snake (Thamnophis proximus proximus) from unknown localities in the state; Haemogregarina spp. from common map turtle (Graptemys geographica) from Marion County; alligator snapping turtle (Macrochelys temminckii) from Ouachita County; C. serpentina from Lincoln County; southern painted turtle (Chrysemys dorsalis) from Pulaski County; Mississippi mud turtle (Kinosternon subrubrum hippocrepis) from Ouachita County; eastern cooter (Pseudemys concinna concinna) from Perry County; and stinkpots (Sternotherus odoratus) from Desha and Lincoln counties; Hepatozoon spp. from western cottonmouth (Agkistrodon piscivorus leucostoma) from Lincoln County; and T. p. proximus from Independence County. Additional surveys on various vertebrates that also include molecular analyses of their intraerythrocytic parasites will surely increase our knowledge of the hematozoans of Arkansas.
For additional information:
Allen, K. E., Michael J. Yabsley, E. M. Johnson, M.V. Reichard, R. J. Panciera, E. A. Ewing, and Susan E. Little. “Novel Hepatozoon in Vertebrates from the Southern United States.” Journal of Parasitology 97 (2011): 648–653.
Barta, John R. “The Dactylosomatidae.” Advances in Parasitology 30 (1991): 1–37.
Barta, John R., and Sherwin S. Desser. “Blood Parasites of Amphibians from Algonquin Park, Ontario.” Journal of Wildlife Diseases 20 (1984): 180–189.
Barta, John R., J. D. Ogedengbe, D. S. Martin, and T. G. Smith. “Phylogenetic Position of the Adeleorinid Coccidia (Myxozoa, Apicomplexa, Coccidia, Eucoccidiorida, Adeleorina) Inferred Using 18S rDNA Sequences.” Journal of Eukaryotic Microbiology 59 (2012): 171–180.
Cook, C. A., S. P. Lawton, A. J. Davies, and N. J. Smith. “Reassignment of the Land Tortoise Haemogregarine Haemogregarina fitzsimonsi Dias 1953 (Adeleorina: Haemogregarinidae) to the Genus Hepatozoon Miller 1908 (Adeleorina: Hepatozoidae) Based on Parasite Morphology, Life Cycle and Phylogenetic Analysis of 18S rDNA Sequence Fragments.” Parasitology 13 (2014): 1–10.
Daly, James J., R. C. McDaniel, J. W. Townsend, and Charles H. Calhoun Jr. “Alterations in the Plasma Membranes of Hepatozoon-Infected Snake Erythrocytes as Evidenced by Differential Staining.” Journal of Parasitology 70 (1984): 151–153.
Davies, A. J., and M. R. L. Johnston. “The Biology of Some Intraerythrocytic Parasites of Fishes, Amphibians and Reptiles.” Advances in Parasitology 45 (200): 1–107.
Davis, A. K., and S. C. Sterrett. “Prevalence of Haemogregarine Parasites in Three Freshwater Turtle Species in a Population in Northeast Georgia, USA.” International Journal of Zoological Research 7 (2011): 156–163.
Desser, Sherwin S. “A Description of Intraerythrocytic Schizonts and Gametocytes of a Haemogregarine of the Snapping Turtle Chelydra serpentine.” Canadian Journal of Zoology 52 (1993): 431–433.
Ernst, C. H., and E. M. Ernst. “Synopsis of Protozoans Parasitic in Native Turtles of the United States.” Bulletin of the Maryland Herpetological Society 15 (1979): 1–15.
Garnham, Percy C. C. “A Haemogregarine in Argas brumpti.” Rivista di Parassitologia 15 (1954): 425–435.
Haklová-Kocíková B, A. Hižnanová, I. Majláth, K. Racka, D. J. Harris, G. Földvári, P. Tryjanowski, N. Kokošová, N. Malceková, and V. Majláthová. “Morphological and Molecular Characterization of Karyolysus—A Neglected but Common Parasite Infecting Some European Lizards.” Parasite Vectors 7 (2014): 55.
Hahn, C. W. “The Stages of Haemogregarina stepanovi Danilewsky Found in the Blood of Turtles, with Special Reference to Changes in the Nucleus.” Archives für Protistenkunde 17 (1909): 307–376.
Harris, D. J., I. Damas-Moreira, J. P. Maia, and A. Perera. “First Report of Hepatozoon (Apicomplexa: Adeleorina) in Caecilians, with Description of a New Species.” Journal of Parasitology 100 (2014): 117–120.
Hull, R.W., and J. H. Camin. “Haemogregarines in Snakes: The Incidence and Identity of the Erythrocytic Stages.” Journal of Parasitology 46 (1960): 515–523.
Karadjian Grégory, Jean-Marc Chavatte, and Irène Landau. “Systematic Revision of the Adeleid Haemogregarines, with Creation of Bartazoon n. g., Reassignment of Hepatozoon argantis Garnham, 1954 to Hemolivia and Molecular Data on Hemolivia stellate.” Parasite 22 (2015): 31.
Khan, R. A., Donald J. Forrester, T. M. Goodwin, and C. A. Ross. “A Haemogregarine from the American alligator (Alligator mississippiensis).” Journal of Parasitology 66 (1980): 324–332.
Kvicerova, Jana, Václav Hypsa, Nela Dvorákova, Peter Mikul?cek, David Jandzik, Michael G. Gardner, Hossein Javanbakht, Ghoulem Tiar, and Pavel Široky. “Hemolivia and Hepatozoon: Haemogregarines with Tangled Evolutionary Relationships.” Protist 165 (2014): 688–700.
Lainson, Ralph, M. C. De Souza, and C. M. Franco. “Natural and Experimental Infection of the lizard Ameiva with Hemolivia stellata (Adeleina: Haemogregarinidae) of the Toad Bufo marinus.” Parasite 14 (2007): 323–328.
Langmann, G. “On Hemosporidia in American Reptiles and Batrachians.” New York Medical Journal 69 (1899): 1–6.
Leveille, A. N., Mosun Ogedengbe, Mian Hafeez, H. H. Tu, and John R. Barta. “The Complete Mitochondrial Genome Sequence of Hepatozoon catesbianae (Apicomplexa; Coccidia; Adeleorina), a Blood Parasite of the Green Frog, Lithobates (Rana) clamitans.” Journal of Parasitology 100 (2013): 651–656.
MacCallum, W. G. “On the Haematozoan Infections of Birds.” Journal of Experimental Medicine 3 (1897): 117–136.
Manwell, R. D. “The Genus Dactylosoma.” Journal of Protozoology 11 (1964): 526–530
McAllister, Chris T. “Hematozoa (Apicomplexa: Haemogregarinidae, Hepatozoidae) from Two Turtles (Testudines: Chelydridae, Emydidae) and Two Snakes (Ophidia: Colubridae, Viperidae), in Southeastern Oklahoma.” Proceedings of the Oklahoma Academy of Science 95 (2015): 119–124.
McAllister, Chris T., Charles R. Bursey, Henry W. Robison, Matthew B. Connior, and Michael A. Barger. “Haemogregarina sp. (Apicomplexa: Haemogregarinidae), Telorchis attenuata (Digenea: Telorchiidae) and Neoechinorhynchus emydis (Acanthocephala: Neoechinorhynchidae) from Map Turtles (Graptemys spp.), in Northcentral Arkansas.” Journal of the Arkansas Academy of Science 68 (2014): 154–157. Online at: https://scholarworks.uark.edu/cgi/viewcontent.cgi?article=1240&context=jaas (accessed February 8, 2020).
McAllister, Chris T., Matthew B. Connior, Henry W. Robison, Thomas J. Fayton, Renn Tumlison, and Stanley E. Trauth. “Hematozoan Parasites (Apicomplexa: Kinetoplastida) of Seven Arkansas Reptiles (Testudines, Ophidia).” Journal of the Arkansas Academy of Science 70 (2016): 273–278. Online at: https://scholarworks.uark.edu/cgi/viewcontent.cgi?article=2216&context=jaas (accessed February 8, 2020).
McAllister, Chris T., and Anthony W. King. “Hemogregarines in the Red-Eared Slider, Chrysemys scripta elegans (Wied) from Arkansas.” Proceedings of the Arkansas Academy of Science 34 (1980): 124. Online at: https://scholarworks.uark.edu/cgi/viewcontent.cgi?article=2677&context=jaas (accessed February 8, 2020).
McAllister, Chris T., Steve J. Upton, and Stanley E. Trauth. “Hemogregarines (Apicomplexa) and Falcaustra chelydrae (Nematoda) in an Alligator Snapping Turtle, Macroclemys temminckii (Reptilia: Testudines), from Arkansas.” Journal of the Helminthological Society of Washington 62 (1995): 74–77.
McAuliffe, J. R. “Hypothesis Explaining Variations of Hemogregarine Parasitemia in Different Aquatic Turtle Species.” Journal of Parasitology 63 (1977): 580–581.
Paterson, W. B., and Sherwin S. Desser. “Observations of Haemogregarina balli sp. n. from the Common Snapping Turtle, Chelydra serpentine.” Journal of Protozoology 23 (1976): 294–301.
Roudabush, Robert L., and G. R. Coatney. “On Some Blood Protozoa of Reptiles and Amphibians.” Transactions of the American Microscopical Society 56 (1937): 291–297.
Siddall, Mark E. “Phylogeny of Adeleid Blood Parasites with a Partial Systematic Revision of the Haemogregarine Complex.” Journal of Eukaryotic Microbiology 42(1995): 116–125.
Siddall, Mark E., and Sherwin S. Desser. “Merogonic Development of Haemogregarina balli (Apicomplexa: Adeleina: Haemogregarinidae) in the Leech Placobdella ornata (Glossiphoniidae), its Transmission to a Chelonian Intermediate Host and Phylogenetic Implications.” Journal of Parasitology 77 (1991): 426–436.
———. “Transmission of Haemogregarina balli from Painted Turtles to Snapping Turtles through the Leech Placobdella ornate.” Journal of Parasitology 87 (2001): 1217–1218.
Smallridge, C. J., and C.M. Bull. “Transmission of the Blood Parasite Hemolivia mariae Between its Lizard and Tick Hosts.” Parasitology Research 85 (1999): 858–863.
Smith, Todd G. “The Genus Hepatozoon (Apicomplexa: Adeleina).” Journal of Parasitology 82 (1996): 565–585.
Smith, Todd G., and Sherwin S. Desser. “Phylogenetic Analysis of the Genus Hepatozoon Miller, 1908 (Apicomplexa: Adeleorina).” Systematic Parasitology 36 (1997): 213–221.
Smith, Todd G., Sherwin S. Desser, and David S. Martin. “The Development of Hepatozoon sipedon sp. nov. (Apicomplexa: Adeleina: Hepatozoidae) in its Natural Host, the Northern Water Snake (Nerodia sipedon sipedon), in the Culicine Vectors Culex pipiens and C. territans, and in an Intermediate Host, the Northern Leopard Frog (Rana pipiens).” Parasitology Research 1994: 559–568.
Telford, Sam R., Jr. Hemoparasites of the Reptilia: Color Atlas and Text. Boca Raton, FL: CRC Press, 2009.
Telford, Sam R., Jr., J. A. Ernst, A. M. Clark, and J. F. Butler. “Hepatozoon sauritus; A polytopic Hemogregarine in Three Genera and Four Species of Snakes in Northern Florida, with Specific Identity Verified from Genome Analysis.” Journal of Parasitology 90 (2004): 352–358.
Wozniak, E. J., Sam R. Telford Jr., and G. L. McLaughlin. “Employment of the Polymerase Chain Reaction in the Molecular Differentiation of Reptilian Hemogregarines and its Application to Preventive Zoological Medicine.” Journal of Zoo and Wildlife Medicine 25 (1994): 538–547.
Chris T. McAllister
Eastern Oklahoma State College





Comments
No comments on this entry yet.